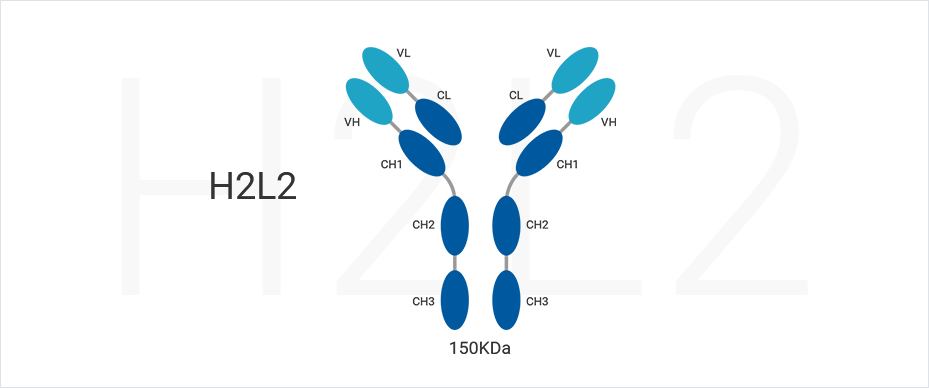
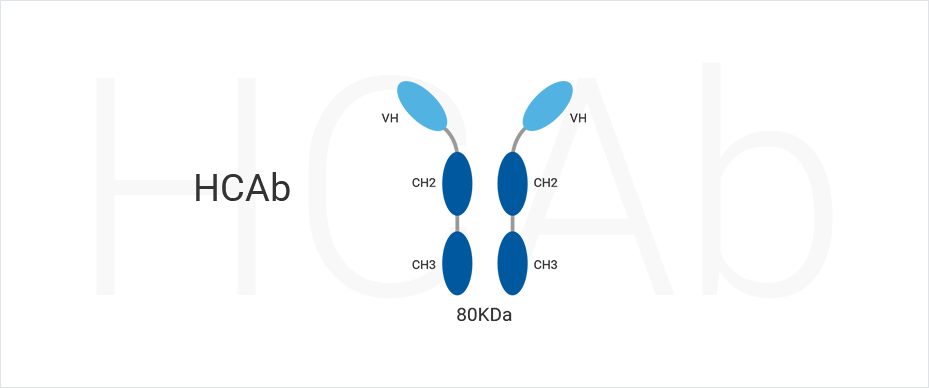
1. Dekker S, Toussaint W, Panayotou G, de Wit T, Visser P, Grosveld F, et al. Intracellularly Expressed Single-Domain Antibody against p15 Matrix Protein Prevents the Production of Porcine Retroviruses. J Virol. 2003;77(22):12132–9. (full text)
2. Janssens R, Dekker S, Hendriks RW, Panayotou G, van Remoortere A, San JK, et al.Generation of heavy-chain-only antibodies in mice. Proc Natl Acad Sci U S A. 2006;103(41):15130–5.(full text)
3. Laventie B-J, Rademaker HJ, Saleh M, de Boer E, Janssens R, Bourcier T, et al. Heavy chain-only antibodies and tetravalent bispecific antibody neutralizing Staphylococcus aureus leukotoxins. Proc Natl Acad Sci U S A. 2011;108(39):16404–9. (full text)
4. Drabek D, Janssens R, de Boer E, et al. Expression Cloning and Production of Human Heavy-Chain-Only Antibodies from Murine Transgenic Plasma Cells. Front Immunol. 2016;7:619.(full text)